New Delhi, April 1
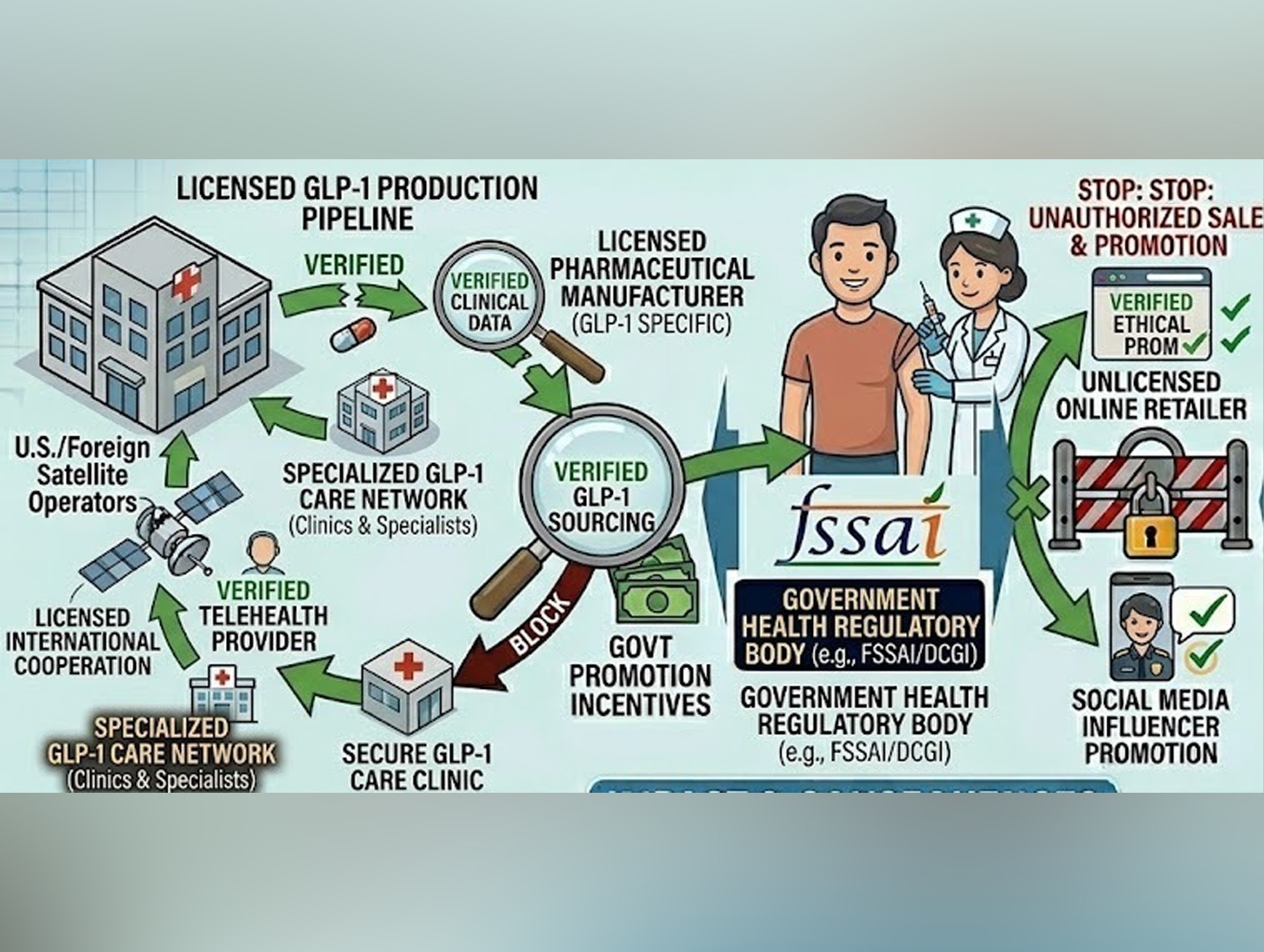
The government on Wednesday cautioned against the unsupervised use of GLP-1-based weight loss drugs, stating that these prescription medications, used to treat type 2 diabetes and obesity, can have serious side effects and must only be taken under the supervision of qualified medical specialists.
The warning comes amid growing concerns over the increasing availability of multiple variants of GLP-1 drugs in the Indian market. Authorities have flagged their on-demand sale through retail pharmacies, online platforms, wholesalers, and wellness clinics as a cause for concern.
In response, the Central Drugs Standard Control Organisation (CDSCO) has stepped up regulatory surveillance to curb unauthorised sale and promotion of these drugs. The government warned that strict inspections will continue in the coming weeks, and businesses found violating norms could face licence cancellation, fines, and legal action.
GLP-1 (glucagon-like peptide-1 receptor agonists) drugs are designed to regulate blood sugar levels and appetite by stimulating insulin release and suppressing excess glucagon. They also slow gastric emptying, increasing the feeling of fullness and aiding in weight loss.
However, officials stressed that misuse of these drugs without proper medical oversight can lead to serious health complications. Side effects can range from nausea and vomiting to severe conditions such as pancreatitis, kidney injury, and bowel obstruction.
The government reiterated that in India, GLP-1 drugs can only be prescribed by specialists such as endocrinologists, internal medicine experts, and cardiologists, and are not available over the counter.
To tackle misuse and misleading promotion, the Drug Controller General of India, in coordination with state drug regulators, has initiated multiple enforcement measures. On March 10, an advisory was issued to manufacturers, directing them to halt misleading advertisements and refrain from promoting the drugs for non-prescription use.
In recent weeks, authorities have conducted inspections of 49 businesses nationwide, including online pharmacy warehouses, wholesalers, retailers, and weight loss clinics. These checks focused on identifying violations related to unauthorised sales, improper prescription practices, and deceptive marketing. Notices have been issued to entities found breaching regulations.
The government emphasised that while GLP-1 drugs represent a significant advancement in treating diabetes and obesity, their safe use requires strict adherence to medical guidance.